One validated platform, total compliance.







Traditional compliance is a bottleneck.



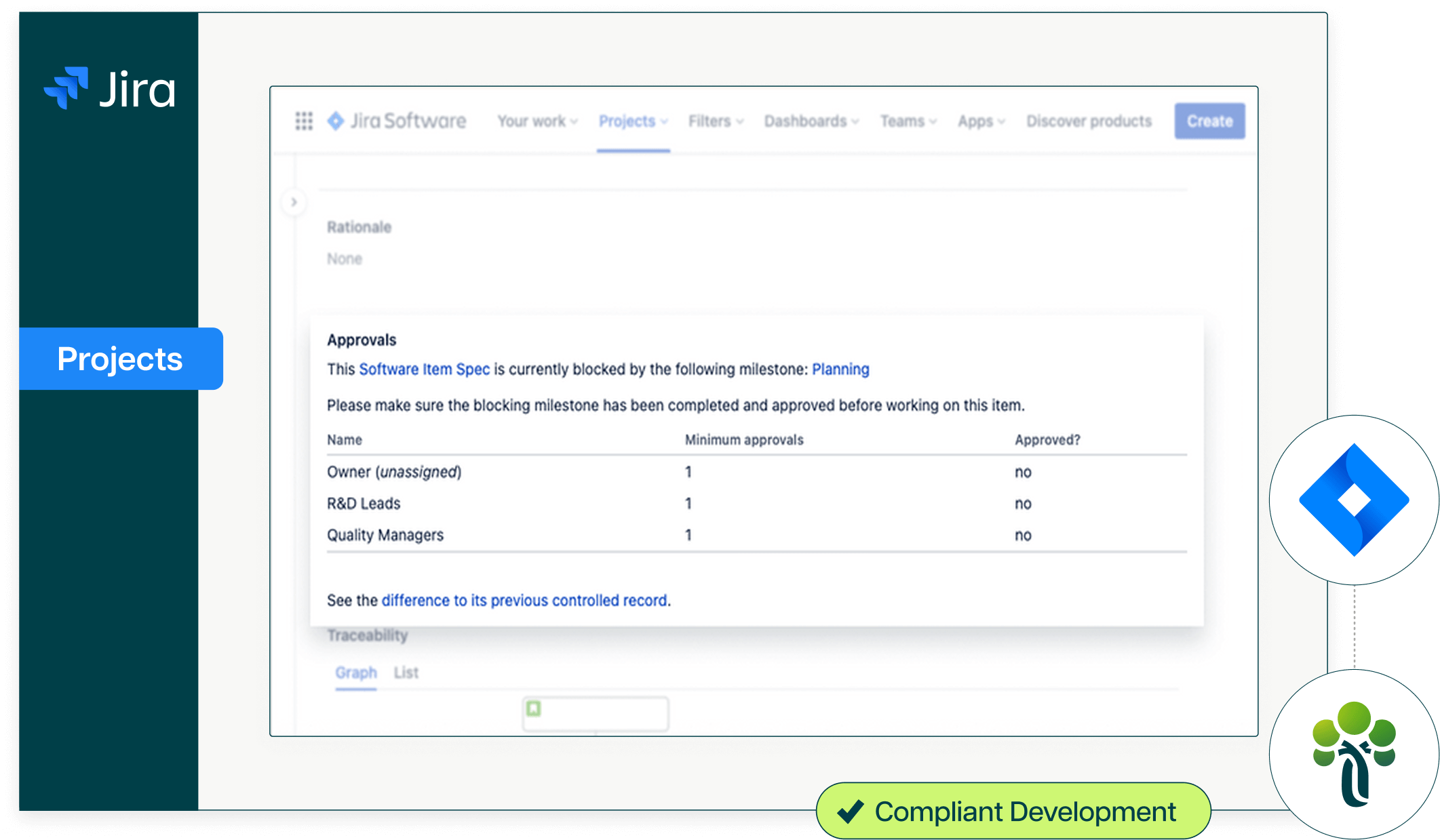
One platform that connects regulatory compliance to development.
Aligned with CSA and GAMP 5
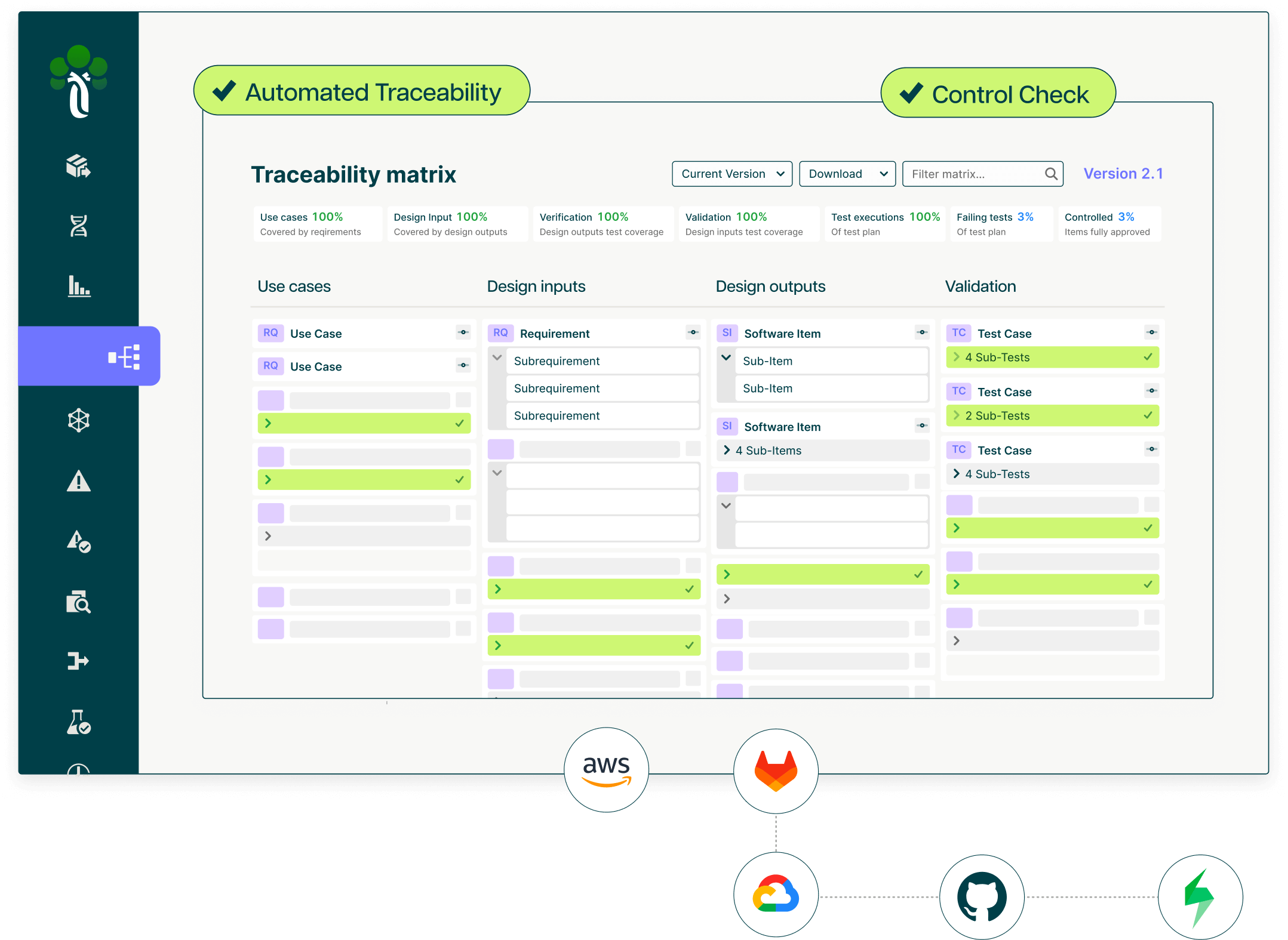
Ketryx replaces static validation documents with living, auto-generated records. Requirements, risks, test protocols, and results are captured, traced, and versioned automatically, supporting URS, FS, DS, IQ, OQ, and PQ documentation aligned with FDA CSA (February 2026) and GAMP 5 2nd Edition, with structured validation evidence always ready to support BLA and NDA submission preparation.
ALCOA+ enforced at every touchpoint
Ketryx enforces ALCOA+ at every touchpoint, automatically attributing, timestamping, and immutably storing every change, approval, and test result across GLP, GMP, and GCP systems. Electronic signatures support 21 CFR Part 11 and Annex 11 with multi-factor authentication, full versioning, and a complete, defensible audit trail.
Connected workflows so nothing falls through the cracks
Ketryx connects change control and CAPA workflows natively, automatically linking Change Requests to affected systems, validation records, risks, and test cases. CAPA traces from root cause to effectiveness check, with instant notification to responsible owners when follow-up validation activity is required.
.png)
.png)
Supplier qualification, SBOMs, and vulnerability management
Pharma and biotech organizations depend on complex vendor, cloud, and third-party software ecosystems. Ketryx manages supplier qualification, vendor testing evidence, SBOM generation, and software inventory in one place. Vulnerability scanning runs continuously against GHSA, with NVD available on request, and supplier records link directly to your system inventory. Forward compatible with EU GMP Annex 22 AI system requirements, subject to final regulatory guidance.
Trusted by 4 of the Top 5 Life Science Leaders
Connects to the tools your team already uses.
Ketryx overlays your existing development and compliance tools — no rip-and-replace required. Your engineers stay in the tools they already use.


How Aignostics is Accelerating AI Development in Pathology with Ketryx
Aignostics is a global artificial intelligence (AI) company that turns complex multi-modal pathology data into transformative insights. With an expanding portfolio of AI-powered pathology products, Aignostics needed a streamlined, integrated solution to support their regulated software development processes and ISO 13485 certification. By transitioning to Ketryx, Aignostics was able to cut their release time down to three months while staying compliant.

.png)